Sino Biopharm Unit Licenses Blood Cancer Drug to Sanofi for Up to $1.53 Billion - Reuters
Sino Biopharm unit licenses blood cancer drug to Sanofi for up to $1.53 billion Reuters
9 stories found
Sino Biopharm unit licenses blood cancer drug to Sanofi for up to $1.53 billion Reuters
Teva and Sanofi announced durable phase 2b efficacy results for their drug duvakitug, intended for the treatment of ulcerative colitis and Crohn's disease.
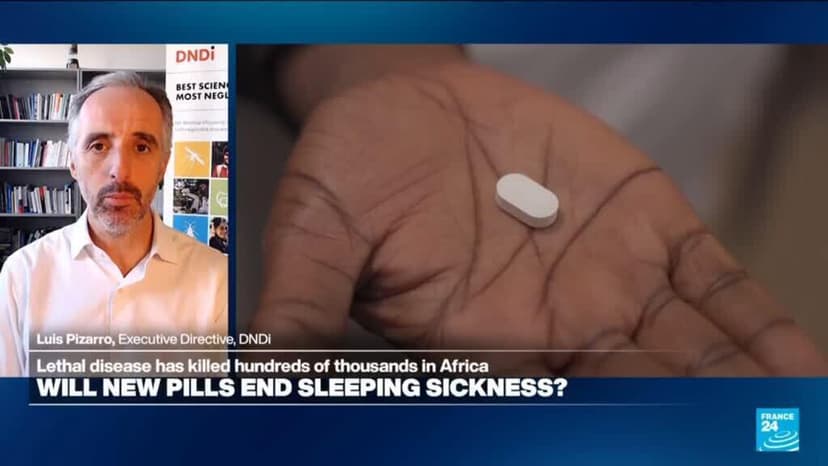
European drug regulators on Friday endorsed a new and simpler treatment for sleeping sickness, in what could be a giant boost to efforts to eliminate the disease. A European Medicines Agency committee gave its nod to acoziborole, developed by Sanofi and DNDi. Speaking to France 24, Dr Luis Pizarro, executive director of DNDi, said the new pill could revolutionise treatment of the disease, which has killed hundreds of thousands across Africa.
Sanofi has announced the appointment of Manuela Buxo as the new head of its Specialty Care Global Business Unit.
Multiple biotech and pharmaceutical companies, including Legend Biotech, Corcept Therapeutics, and Ultragenyx, have released updates on their drug pipelines, clinical trial data, and regulatory interactions, impacting market outlook.
Regeneron and Sanofi have announced that their drug Dupixent has received a label expansion, allowing it to be used for the treatment of allergic fungal rhinosinusitis.
Texas sues Sanofi for allegedly 'bribing' providers to boost drug prescriptions Reuters
Drugmaker Sanofi announced plans to expand its India Global Capability Center, aiming to increase its workforce to over 4,500 employees.